Experiment
TD1000

IDEAL GASES - BOYLE'S LAW
Bench top apparatus that demonstrates the relationship between pressure and volume of an ideal gas at a fixed temperature.
If you have any questions or you'd like to discuss a product, please call us.
+44 1159 722 611IDEAL GASES - BOYLE'S LAW
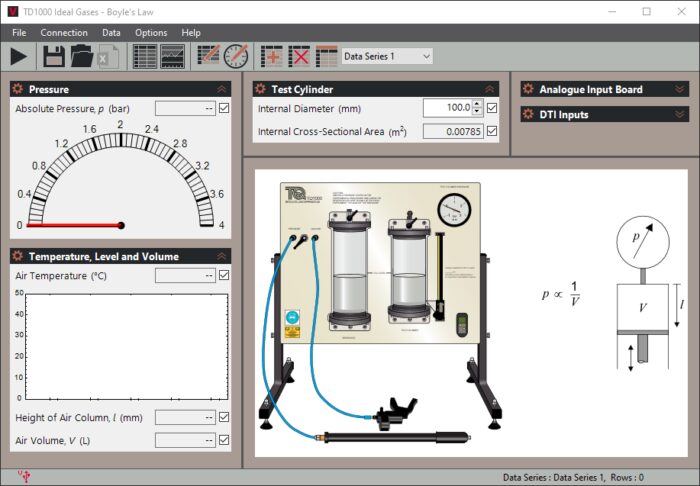
The bench-mounting equipment includes a back plate that holds two clear-walled cylinders containing oil (supplied). Students use hand-operated pumps (supplied) to increase or decrease the pressure in the left-hand cylinder (the Reservoir) which moves a ‘liquid piston’ of oil in the righthand cylinder (the test cylinder). This piston compresses or decompresses a trapped column of air in the test cylinder. The equipment uses normal, clean, dry air, as it behaves as an ideal gas over the range of pressures used in this equipment.
A digital indicator measures the change in height of the trapped air column. When multiplied by the cross-sectional area of the column, this gives the change in volume. A mechanical pressure gauge measures the pressure of the trapped air.
A thermocouple and digital display measure the temperature of the trapped air to make sure that students maintain a constant air temperature during tests. They also help to demonstrate the change in air temperature during demonstrations.
Students maintain a constant temperature while recording the changes in volume with applied pressure. They then plot the results to prove Boyle’s Law.
Users can do tests with or without a computer connected. However, for quicker tests with easier recording of results, TecQuipment can supply the optional Versatile Data Acquisition System (VDAS®). This gives accurate real-time data capture, monitoring and display, calculation and charting of all the important readings on a computer.
For connection to VDAS® the apparatus includes an electronic pressure transducer, a thermocouple amplifier and a lead to connect the digital indicator to VDAS®.
Learning outcomes
- Demonstrations of gas temperature change during compression and decompression.
- Proving Boyle’s Law by experiment.